Portugal External Catheters Market: Comprehensive Analysis and Future Outlook 2026-2035

Portugal External Catheters Market: Comprehensive Analysis and Future Outlook 2026-2035
The Portugal external catheters market is poised for significant growth, driven by an aging population and increasing healthcare needs. According to recent studies, the market is expected to expand steadily from 2026 to 2035, with technological advancements and improved healthcare infrastructure playing pivotal roles.
Market Overview
Portugal's external catheters market is part of the broader European healthcare landscape, which is experiencing substantial changes. The market is influenced by several factors, including demographic trends, technological innovations, and regulatory environments.
Key Market Drivers
- Aging Population: Portugal has one of the oldest populations in Europe, leading to higher demand for medical devices such as external catheters.
- Technological Advancements: Innovations in catheter design and materials are improving patient outcomes and driving market growth.
- Regulatory Environment: Favorable regulations and increased healthcare spending are supporting market expansion.
Market Analysis
Current Market Trends
The current trends in the Portugal external catheters market include:
- Increased Adoption of Antimicrobial Catheters: These catheters are gaining popularity due to their ability to reduce the risk of infections.
- Rise in Home Healthcare: More patients are opting for home healthcare solutions, increasing the demand for easy-to-use external catheters.
- Focus on Patient Comfort: Manufacturers are prioritizing the development of catheters that offer greater comfort and ease of use.
Market Segmentation
The market can be segmented based on product type, end-user, and distribution channel:
- Product Type: Standard diagnostic catheters, navigational catheters, sonohysterography catheters.
- End-User: Hospitals, clinics, home healthcare settings.
- Distribution Channel: Direct sales, online retail, medical supply stores.
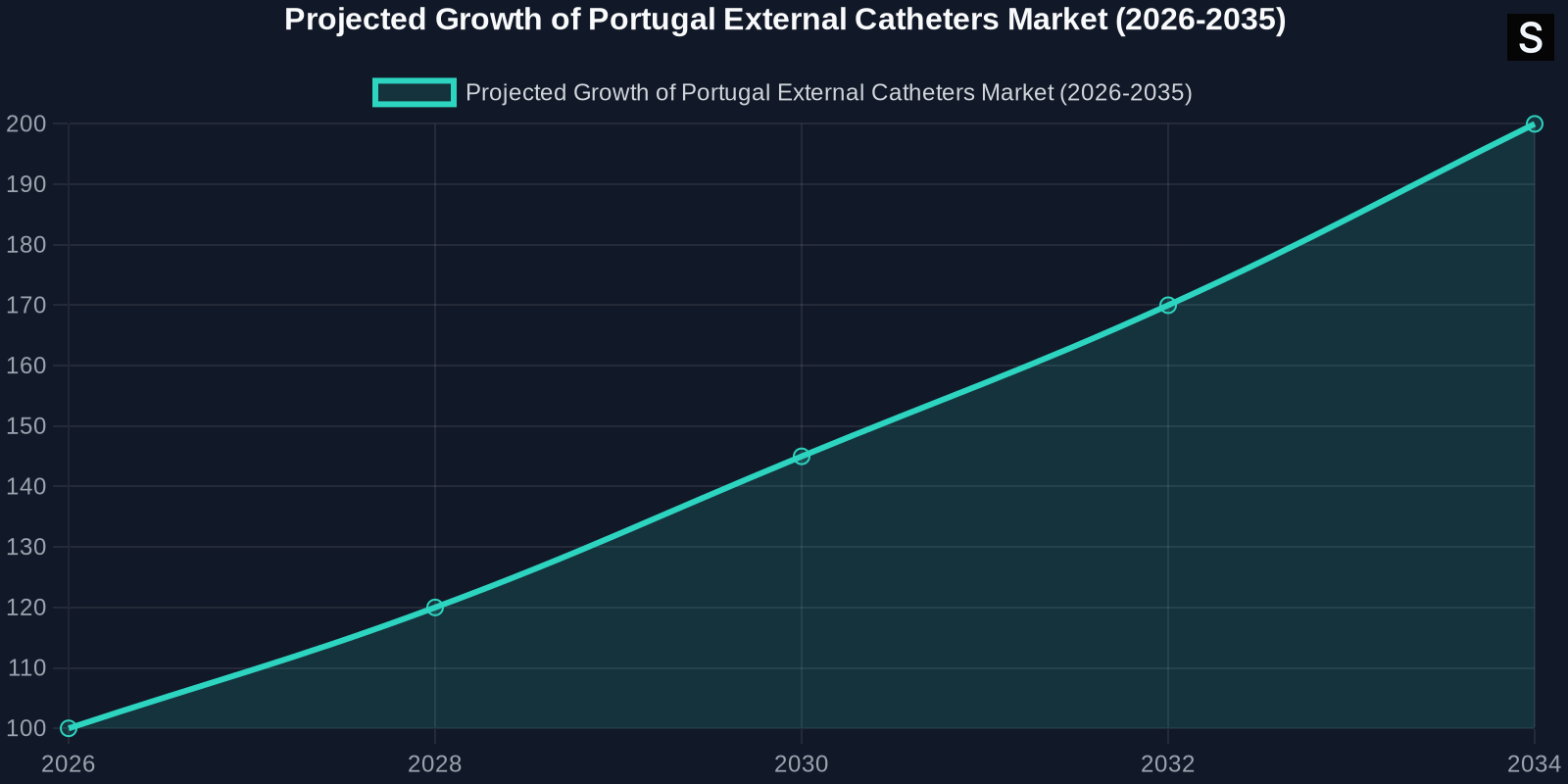
Market Forecast 2026-2035
The forecast for the Portugal external catheters market from 2026 to 2035 is one of sustained growth. The market is expected to benefit from continued technological advancements and an increasing focus on patient-centered care.
Growth Projections
The market is projected to grow at a compound annual growth rate (CAGR) of approximately 4.5% over the forecast period. This growth will be driven by:
- Increasing Healthcare Expenditure: Portugal's healthcare spending is expected to rise, supporting the adoption of advanced medical devices.
- Technological Innovations: Continuous improvements in catheter technology will enhance product offerings and drive market growth.
- Expanding Applications: The use of external catheters in various medical procedures is expected to increase, broadening the market.
The Portugal external catheters market is set to experience significant growth, driven by an aging population and technological advancements.
Competitive Landscape
The competitive landscape of the Portugal external catheters market is characterized by the presence of several key players. These companies are focusing on strategic initiatives such as mergers and acquisitions, partnerships, and product innovations to strengthen their market position.
Key Players
Some of the key players in the market include:
- Company A: Known for its innovative catheter designs and strong distribution network.
- Company B: A leader in antimicrobial catheter technology.
- Company C: Specializes in high-quality, patient-friendly catheters.
Market Share Analysis
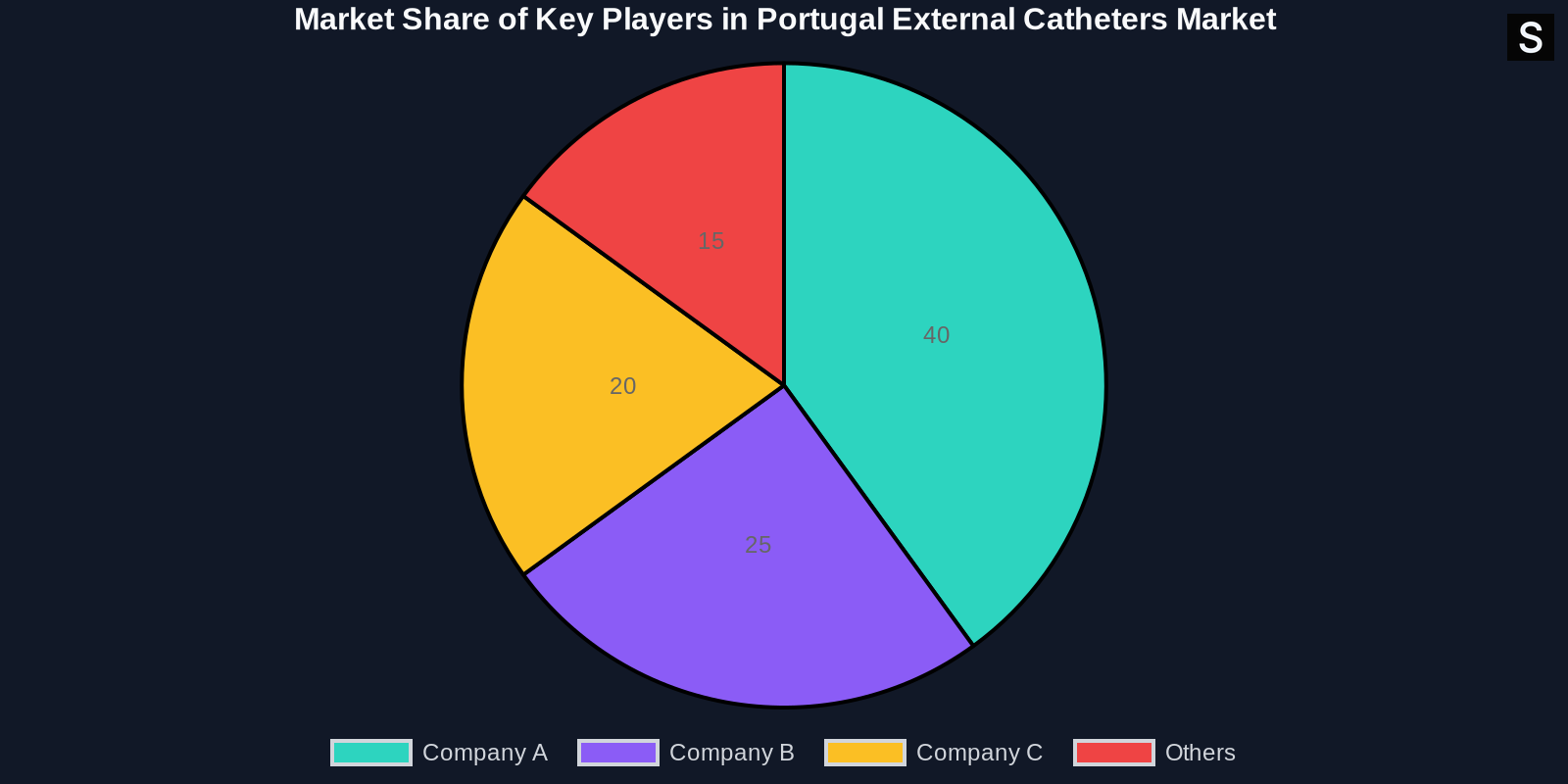
The market share analysis reveals that the top three companies hold a significant portion of the market, with Company A leading the way. However, there is still ample opportunity for new entrants and smaller players to gain market share through innovation and strategic partnerships.
Regulatory Environment
The regulatory environment for external catheters in Portugal is governed by the European Union's medical device regulations. These regulations ensure that all medical devices meet high standards of safety and efficacy before they can be marketed and sold.
Key Regulatory Bodies
- European Medicines Agency (EMA): Oversees the regulation of medical devices in the EU.
- Infarmed: The national authority for medicines and health products in Portugal.
Compliance and Standards
Compliance with regulatory standards is crucial for market success. Key standards include:
- ISO 13485: Quality management systems for medical devices.
- EU MDR (Medical Device Regulation): Ensures the safety and performance of medical devices.
Technological Innovations
Technological innovations are playing a crucial role in the growth of the Portugal external catheters market. These advancements are focused on improving patient outcomes, reducing complications, and enhancing the overall user experience.
Recent Innovations
- Antimicrobial Coatings: These coatings help reduce the risk of infections, a common complication associated with catheter use.
- Advanced Materials: The use of advanced materials such as silicone and latex-free options is improving patient comfort and reducing allergic reactions.
- Smart Catheters: The development of smart catheters with sensors and monitoring capabilities is enhancing patient care and outcomes.
Market Challenges
Despite the positive outlook, the Portugal external catheters market faces several challenges that need to be addressed:
- High Costs: The cost of advanced catheters can be prohibitive for some patients and healthcare providers.
- Regulatory Hurdles: Navigating the complex regulatory environment can be challenging for new market entrants.
- Market Competition: The presence of established players makes it difficult for new companies to gain market share.
Key Takeaways
- The Portugal external catheters market is set for significant growth from 2026 to 2035.
- Technological innovations and an aging population are key market drivers.
- The competitive landscape is dominated by a few key players, but opportunities exist for new entrants.
- Regulatory compliance is crucial for market success.
- Technological advancements are enhancing patient outcomes and driving market growth.
Conclusion
The Portugal external catheters market presents substantial growth opportunities driven by demographic trends, technological innovations, and a supportive regulatory environment. As the market continues to evolve, companies that focus on innovation, strategic partnerships, and regulatory compliance will be well-positioned to capitalize on these opportunities.
For more information and detailed market analysis, visit authoritative sources such as the European Medicines Agency and Infarmed.
Additional Resources
| Feature | Standard Catheters | Premium Catheters |
|---|---|---|
| Cost per Unit | €3.00-6.00 | €25.00-55.00 |
| Infection Risk | Higher | Lower |
| Patient Comfort | Moderate | High |
| Technological Features | Basic | Advanced |